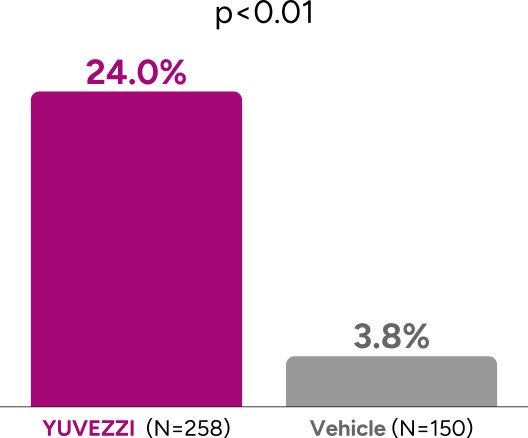
Primary endpoint: Significantly more patients achieved ≥3-line gain BUCNVA without ≥1-line loss in BUCDVA with YUVEZZI than with vehicle or monotherapies1
BUCNVA = binocular uncorrected near visual acuity; BUCDVA = binocular uncorrected distance visual acuity.
The efficacy of YUVEZZI was rigorously evaluated
YUVEZZI was studied in over 600 presbyopia patients (healthy males and females aged 45 to 80) across two Phase 3 randomized, double-masked, controlled studies, BRIO I and BRIO II.1
Significant and sustained functional near vision improvement
Functional Near Vision
~70%
Nearly 70% of patients achieved 20/40 or better near visual acuity at Hour 2 with YUVEZZI3*
Reading Speed
6
+
6x greater improvement in reading speed at 3 months with YUVEZZI compared to vehicle3
No evidence of tachyphylaxis with YUVEZZI from the first administration to 12 months3
Tachyphylaxis is a diminished response to successive doses of a drug, rendering it less effective over time.5
*
Proportion of subjects with a Snellen equivalent of 20/40 vision or better (≥68 letters) in mesopic BUCNVA at 40 cm using both eyes at each timepoint (including Hour 0) for YUVEZZI vs vehicle (Days 7, 270, and 360).3