With established long-term safety, your patients can have confidence using YUVEZZI
Most common treatment-related adverse events from
BRIO II (reported in >5% of participants)2
| YUVEZZI (N = 358) | Vehicle (N = 159) | |
|---|---|---|
| Headache | 56 (15.6%) | 1 (0.6%) |
| Eye irritation (upon instillation) | 50 (14.0%) | 1 (0.6%) |
| Eye pain (upon instillation) | 24 (6.7%) | 0 (0.0%) |
| Visual impairment | 23 (6.4%) | 1 (0.6%) |
No overall differences in safety observed between adult and elderly patients1
*
YUVEZZI was evaluated in 536 patients with presbyopia in 2 randomized double-masked controlled studies. BRIO I was a single-dose crossover study in which 178 patients received YUVEZZI. BRIO II included 358 patients who received YUVEZZI in the long-term safety portion of the study (up to 12 months' duration). Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.1
No treatment-related serious adverse reactions through 12 months1*
The majority of adverse reactions were mild, transient, and self-resolving1
No observed retinal detachments through 12 months2
Treatment-related vitreous detachment was similar with YUVEZZI (0.8%) compared to vehicle (0.6%)2
Rare cases of retinal tear and detachment have been reported with miotics. Individuals with pre-existing retinal disease are at increased risk of retinal detachment. Examination of the retina is advised in all patients prior to the initiation of therapy1
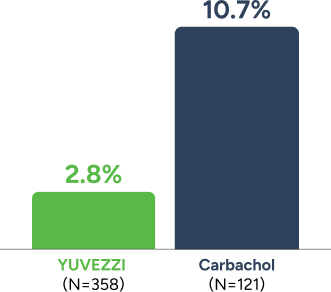
Low treatment-related hyperemia with YUVEZZI2
Patients demonstrated less treatment-related hyperemia with YUVEZZI than with carbachol alone2
Patients experiencing hyperemia